Outbreak of Extensively Drug-resistant Pseudomonas aeruginosa Associated with Artificial Tears
CDC is collaborating with the Food and Drug Administration (FDA) and state and local health departments to investigate a multistate outbreak of an extensively drug-resistant strain of Pseudomonas aeruginosa. The outbreak strain, carbapenem-resistant Pseudomonas aeruginosa with Verona integron-mediated metallo-β-lactamase and Guiana extended-spectrum-β-lactamase (VIM-GES-CRPA), had never been reported in the United States prior to this outbreak. The outbreak is associated with multiple types of infections, including eye infections. The investigation to date has identified artificial tears as a common exposure for many patients.
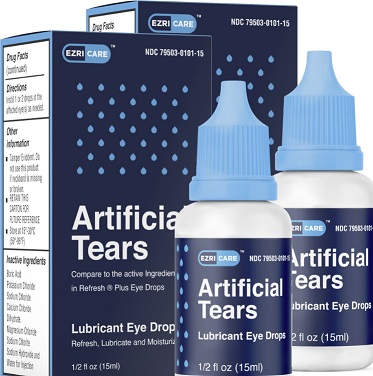
At this time, CDC and FDA recommend clinicians and patients stop using EzriCare or Delsam Pharma’s Artificial Tears products pending additional guidance from CDC and FDA.
Current Update
As of March 1, 2023, CDC, in partnership with state and local health departments, identified 64 patients in 13 states (CA, CO, CT, FL, IL, NJ, NM, NY, NV, TX, UT, WA, WI) with VIM-GES-CRPA, a rare strain of extensively drug-resistant P. aeruginosa. Thirty-seven patients were linked to four healthcare facility clusters. One person has died and there have been 8 reports of vision loss. Dates of specimen collection were from May 2022 to January 2023. Isolates have been identified from clinical cultures of sputum or bronchial wash (15), cornea (14), urine (9), other nonsterile sources (4), and blood (2), and from rectal swabs (26) collected for surveillance; some patients had specimens collected from more than one anatomic site.
Most patients reported using artificial tears. Patients reported over 10 different brands of artificial tears and some patients used multiple brands. EzriCare Artificial Tears, a preservative-free, over-the-counter product packaged in multidose bottles, was the brand most commonly reported. This was the only common artificial tears product identified across the four healthcare facility clusters. Laboratory testing by CDC identified the presence of VIM-GES-CRPA in opened EzriCare bottles from multiple lots; these bottles were collected from patients with and without eye infections and from two states. VIM-GES-CRPA recovered from opened products match the outbreak strain. Testing of unopened bottles of EzriCare Artificial Tears is ongoing to assist in evaluating for whether contamination may have occurred during manufacturing. Patients and healthcare providers should immediately stop the use of EzriCare Artificial Tears pending additional information and guidance from CDC and FDA.
FDA encourages health care professionals and patients to report adverse events or quality problems with any medicine to FDA’s MedWatch Adverse Event Reporting program. Consumers may also report adverse reactions by contacting FDA’s Consumer Complaint Coordinators.
Patient Information
Patients should stop using EzriCare or Delsam Pharma’s Artificial Tears pending additional information and guidance from CDC and FDA. If patients were advised to use EzriCare or Delsam Pharma’s Artificial Tears by their healthcare provider, they should follow up with their healthcare provider for recommendations about alternative treatment options.
Patients who have used EzriCare or Delsam Pharma’s artificial tears and who have signs or symptoms of an eye infection should seek medical care immediately. At this time, there is no recommendation for testing of patients who have used this product and who are not experiencing any signs or symptoms of infection.
Eye infection symptoms may include:
– Yellow, green, or clear discharge from the eye
– Eye pain or discomfort
– Redness of the eye or eyelid
– Feeling of something in your eye (foreign body sensation)
– Increased sensitivity to light
– Blurry vision
FDA encourages health care professionals and patients to report adverse events or quality problems with any medicine to FDA’s MedWatch Adverse Event Reporting program. Consumers may also report adverse reactions by contacting FDA’s Consumer Complaint Coordinators.
The infections are caused by a strain of carbapenem-resistant P. aeruginosa that produces the Verona integron-encoded metallo-β-lactamase (VIM) and the Guiana-Extended Spectrum-β-Lactamase (GES). Isolates are P. aeruginosa sequence type (ST) 1203 and harbor blaVIM-80 and blaGES-9, a combination not previously identified in the United States.
VIM-GES-CRPA isolates associated with this outbreak have been extensively drug-resistant (XDR). Isolates that underwent testing at public health laboratories were not susceptible to cefepime, ceftazidime, piperacillin-tazobactam, aztreonam, carbapenems, ceftazidime-avibactam, ceftolozane-tazobactam, fluoroquinolones, polymyxins, amikacin, gentamicin, and tobramycin. A subset of 3 isolates that underwent antimicrobial susceptibility testing for cefiderocol were susceptible to this agent.
Carbapenem-resistance mechanism testing for carbapenem-resistant Pseudomonas aeruginosa and expanded antimicrobial susceptibility testing for clinical decision making is available through the Antibiotic Resistance Laboratory Network which can be accessed through state health department healthcare-associated infections programs.